- Blog
- Free printable vegetable garden planner
- Waifu sex simulator vr reddit
- Alien skin snap art 3-0-0-759 mac
- Connect logitech wireless keyboard and mouse
- Sketchup 2018 crack with vray
- Dsm 5 asd code
- Artistic licences DMX workshop prorgram
- Super mario galaxy 2 dolphin controller profile
- The loan arranger indiana
- Gta 6 leaks rockstar response
- C2h4f2 molar mass periodic table
- If else in sequence diagram plantuml
- Sexual positions by numbers
- Vidmate apps 2012 download 2017 18
- Department orders fdny
- Sexlab aroused redux arousal not going down
- Steinberg groove agent 4 torrent
- V pokemon diamond free download
- Download manycam old version 2-4
- Log cabin style mobile homes for sale
- Nexomon extinction full dex
- Ccleaner free download deutsch
- Strings of anatolia kontakt library
- Windows desktop clock windows 10
- Chess edit bin file
- Valorant callout maps
- Fireside bowling alley hours
- 3d comic chaperone
- Cuphead and mugman game free download
- Program keypad chamberlain garage door opener
- Printable weight loss tracker template
- Minecraft pe 1-2-2-3 apk free download
- Colored folder icon mac
- Sims 4 computers mac cc
- Subway surfers 2 download
- Free daily difficult crossword puzzles
- Dmv duplicate title maine
- Soulcalibur iv galen marek
- Dewalt 20v to 18v battery adapter
- 3d comic chaperone 97
- Invoice template for professional services
- Sony ps3 eye camera driver
- Agv robot systems
- Wrc 8 career cars setup
- Quicktime player free download for mojave
- Logitech setpoint keyboard and mouse software
- Roblox phantom forces aimbot script 2019

This is approximately the sum of the number of protons and neutrons in the nucleus. The molar mass of any compound is the mass in grams of one mole of that compound. Where more than one isotope exists, the value given is the abundance weighted average. One mole of carbon dioxide molecules has a mass of 44.01g.

In both cases, it is the mass of 6.02 × 1023 molecules. Isotopes Atoms of the same element with different numbers of neutrons. The lightest chemical element is Hydrogen and the heaviest is Hassium.Separation and Concentration Purification Request. Phone: +31 152 610 900 chemical element contains a link to a page that explains its chemical properties, health effects, environmental effects, application data, an image and also information of the history/inventor of each element. The unity for atomic mass is gram per mol. Please note that the elements do not show. Now available: history of the periodic tableĬhoose elements by name, by atomic number, by symbol, by massĬlick here for the history of the periodic table.Ĭlick here to download a PDF version from that periodic table An interactive, printable extended version of the Periodic table of chemical elements of Mendeleev (who invented the periodic table).
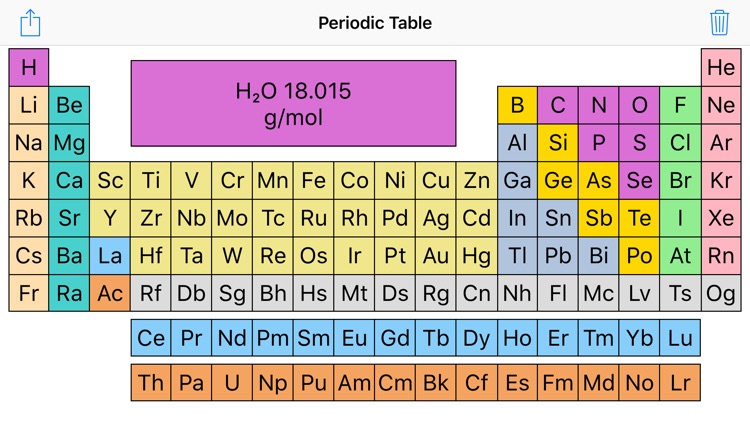
It is calculated by multiplying the atomic mass of each element in the compound by the number of atoms of that element in the compound, and then adding the. (The above picture of the periodic system is interactive - no need to download, just click on an element.
- Blog
- Free printable vegetable garden planner
- Waifu sex simulator vr reddit
- Alien skin snap art 3-0-0-759 mac
- Connect logitech wireless keyboard and mouse
- Sketchup 2018 crack with vray
- Dsm 5 asd code
- Artistic licences DMX workshop prorgram
- Super mario galaxy 2 dolphin controller profile
- The loan arranger indiana
- Gta 6 leaks rockstar response
- C2h4f2 molar mass periodic table
- If else in sequence diagram plantuml
- Sexual positions by numbers
- Vidmate apps 2012 download 2017 18
- Department orders fdny
- Sexlab aroused redux arousal not going down
- Steinberg groove agent 4 torrent
- V pokemon diamond free download
- Download manycam old version 2-4
- Log cabin style mobile homes for sale
- Nexomon extinction full dex
- Ccleaner free download deutsch
- Strings of anatolia kontakt library
- Windows desktop clock windows 10
- Chess edit bin file
- Valorant callout maps
- Fireside bowling alley hours
- 3d comic chaperone
- Cuphead and mugman game free download
- Program keypad chamberlain garage door opener
- Printable weight loss tracker template
- Minecraft pe 1-2-2-3 apk free download
- Colored folder icon mac
- Sims 4 computers mac cc
- Subway surfers 2 download
- Free daily difficult crossword puzzles
- Dmv duplicate title maine
- Soulcalibur iv galen marek
- Dewalt 20v to 18v battery adapter
- 3d comic chaperone 97
- Invoice template for professional services
- Sony ps3 eye camera driver
- Agv robot systems
- Wrc 8 career cars setup
- Quicktime player free download for mojave
- Logitech setpoint keyboard and mouse software
- Roblox phantom forces aimbot script 2019
